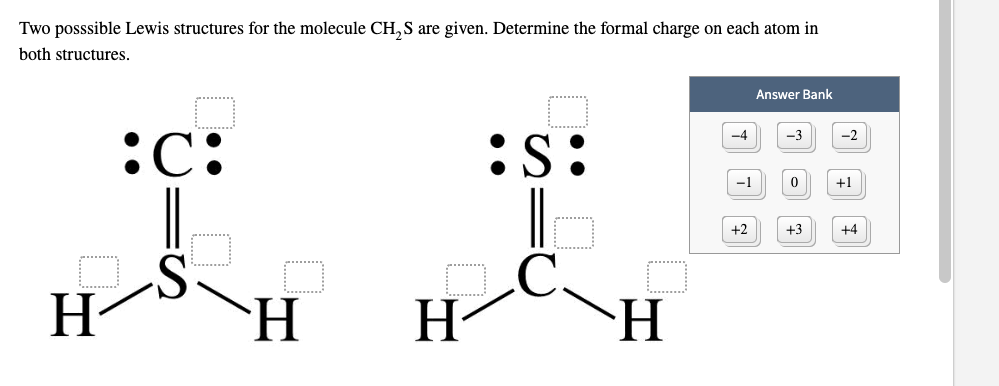 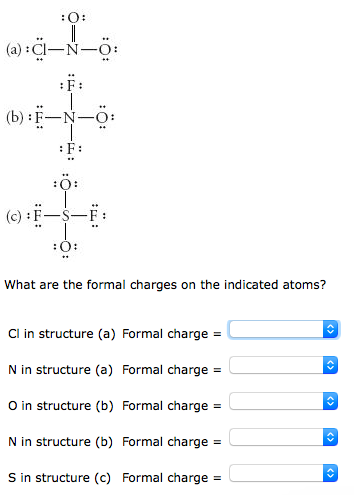 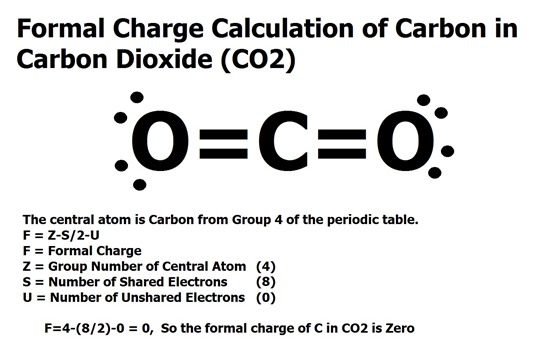
If A and X are in the same group, place the atom with the higher. The calculator will readily determine the formal charge. For molecules of the formula AXn, place the atom with the lower group number in the center. Step 2: Calculate the formal charge of the compound using the Lewis Dot Structure in. valence electrons of each atom in the molecule. Step 1: Draw the Lewis Dot Structure for the compound given in order to calculate the formal charge. Yes, just switch the single and double bonds for the above two structures (with the oxygens and their lone electrons). ADVERTISEMENT Chemistry Calculators Formal Charge Calculator Formal Charge Calculator Enter the number of valence electrons, number of lone and bonded pairs. 2) Calculate the total number of electrons for the Lewis structure by summing the. Each electron counts as one and so a pair counts as two. Lone Pairs lone electrons sitting on the atom. In the left resonance structure, all the atoms have zero formal charge, while on the right structure, the nitrogen has a +1 formal charge, and the oxygen with the single bond has a -1 formal charge.Ĭan you draw two additional resonance structures for the above molecules? Formal Charge valence electrons on neutral atom ( lone electron pairs) + (½ bonding electrons) Valence electrons corresponds to the group number of the periodic table (for representative elements).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
